Clinical
EVIDENCE
IDE Trial Enrollment Completed
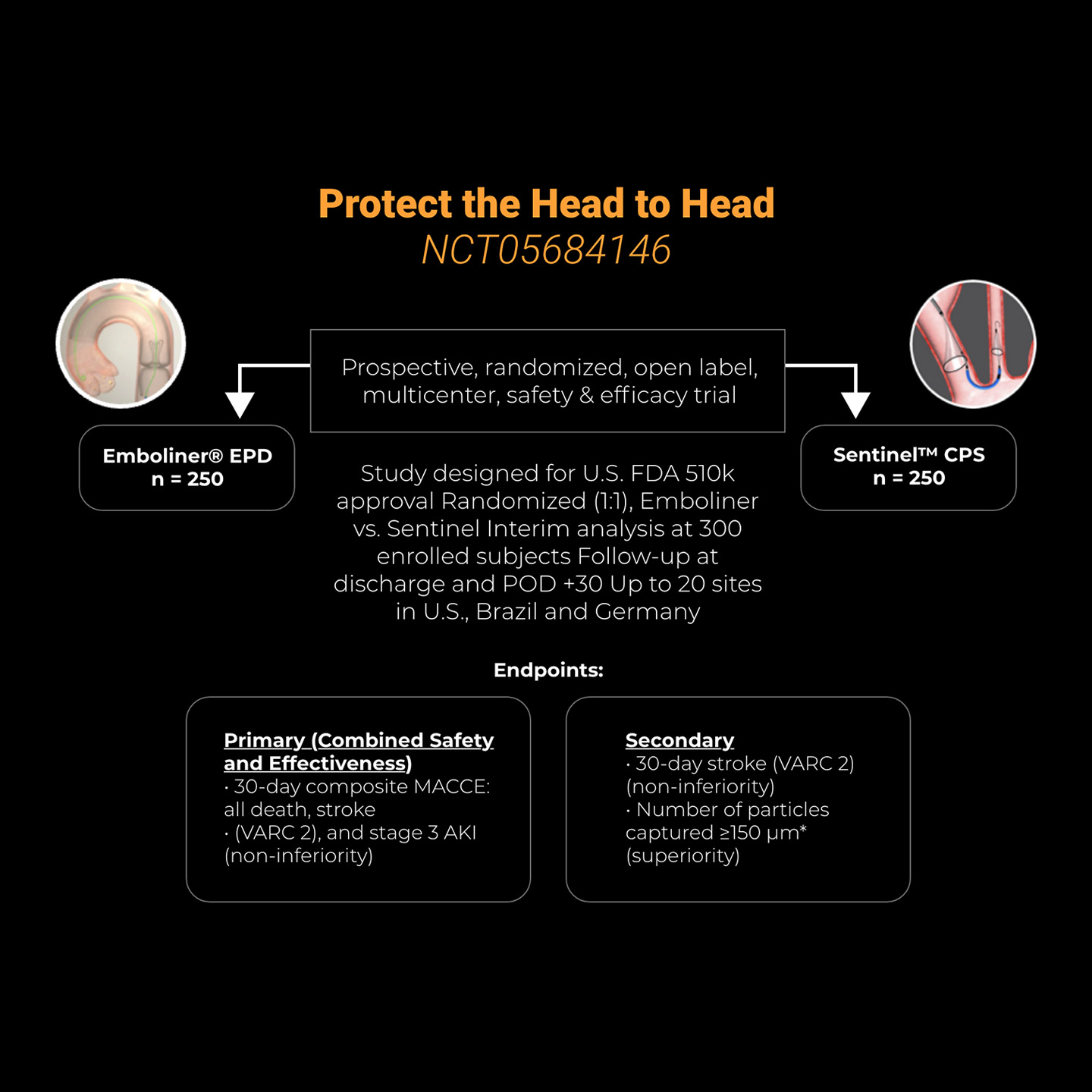
In October 2025, Emboline completed enrollment in the Protect the Head to Head IDE trial (NCT05684146), which is a 500 patient, prospective, randomized, open label, multicenter, 2-arm study to demonstrate safety and effectiveness of the Emboliner® Embolic Protection Catheter compared to a control device (SentinelTM CPS, Boston Scientific) for patients undergoing TAVR. Data from the IDE study will be used to file for FDA commercial approval in the U.S. and CE mark in Europe.
Trial results were presented on March 29, 2026 at ACC by Adam B. Greenbaum MD. The trial demonstrated that the Emboliner is non-inferior to Sentinel in terms of 30-day safety and efficacy endpoints — composite MACCE (death, stroke and stage-3 acute kidney injury) and all stroke, and is superior to Sentinel in terms of debris capture. Although the Emboliner was used for the first time and all operators were experienced Sentinel users, study results demonstrated a higher rate of technical and procedural success, and no significant difference in rate of bleeding or vascular complications between Emboliner and Sentinel.
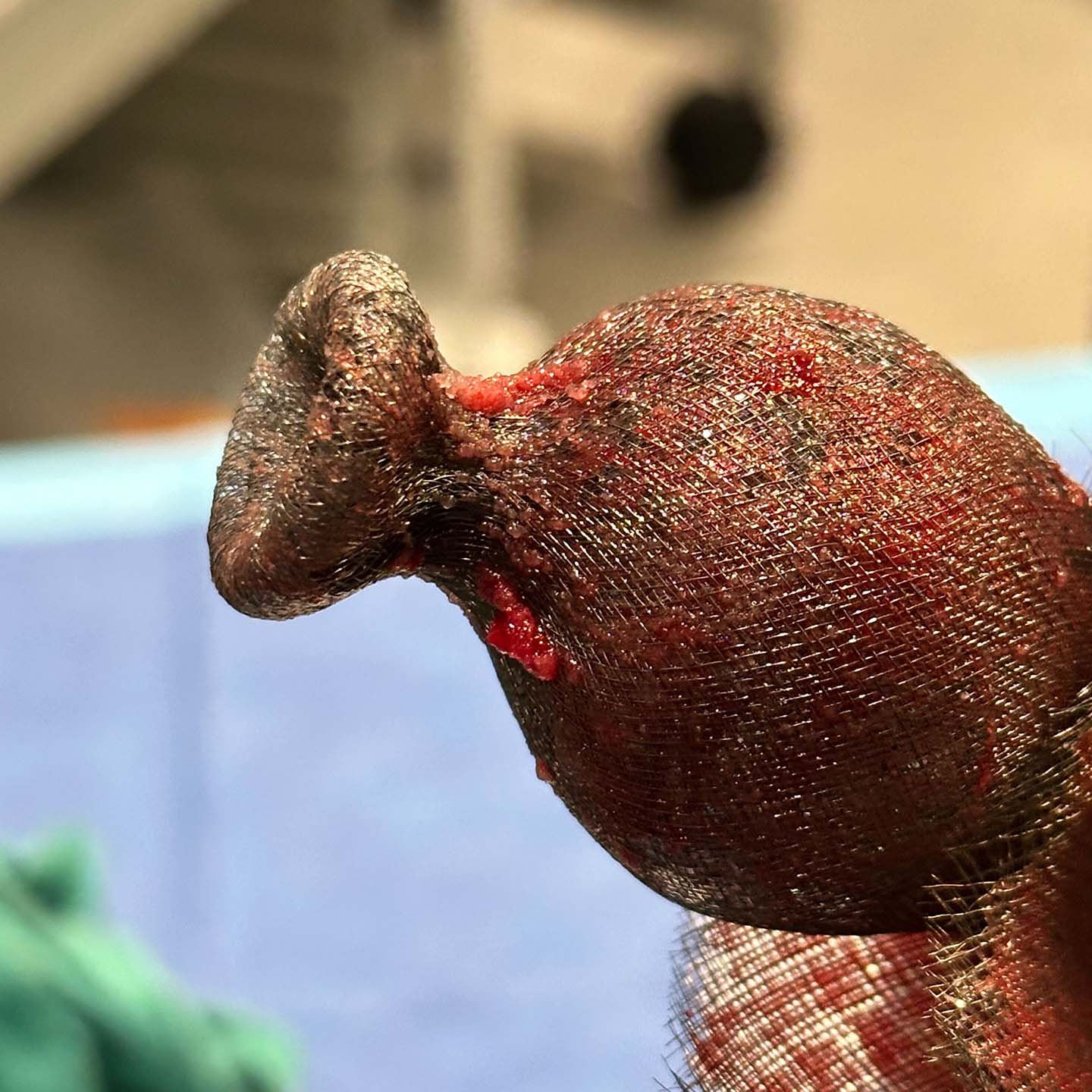
Early Feasibility Studies Demonstrated Safety and Efficacy
In the SafePass series of early feasibility studies (EFS), the Emboliner has been demonstrated to be safe, effective and easy to use, with superior debris capture when compared to historical data. 1
References:
- 1 Grubman D, et al. Predictors of Cerebral Embolic Debris During Transcatheter Aortic Valve Replacement: The SafePass 2 First-in-Human Study. Am J Cardiol. 2023 Nov 15;207:28-34.
